

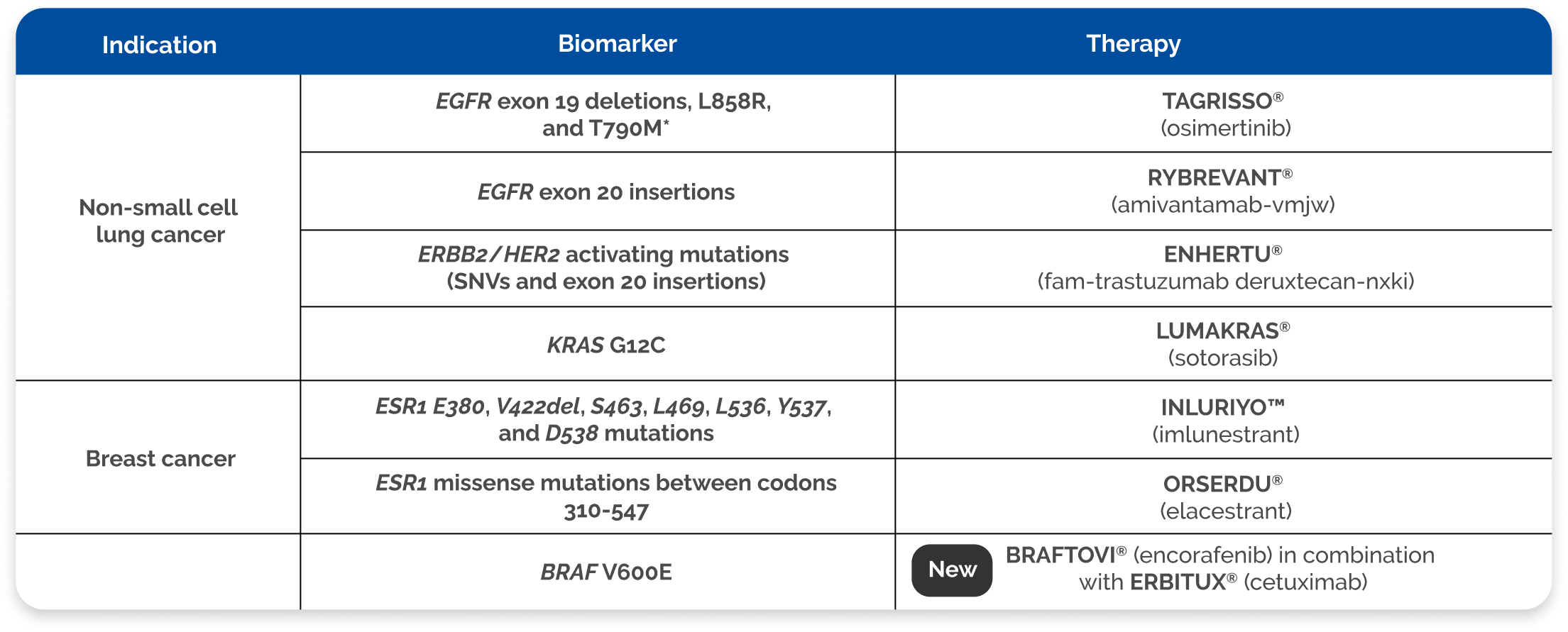
Multiple FDA-approved companion diagnostics approvals across NSCLC, breast cancer, and colorectal cancer1,2
Identify biomarkers to match patients to targeted therapy1,2
Home Oncology Portfolio Partnerships European Policy

References: 1. Guardant360® Liquid Assay Specifications. 2025. Guardant Health, Inc. Redwood City, CA. 2. Guardant Health data on file. April 3, 2025. Guardant Health, Inc. Redwood City, CA.

Multiple FDA-approved companion diagnostics approvals across NSCLC, breast cancer, and colorectal cancer1,2

Home Oncology Portfolio Partnerships European Policy